“The results show that the Favipiravir treatment deserves a more in depth monotherapeutic study of patients with medium or high viremia” explains Dr. Abdul Bing, ALIMA’s medical team leader in Guinea.
The data also shows the frequency of renal dysfunction in patients and suggest that the trials should take into account the initial viral load value.
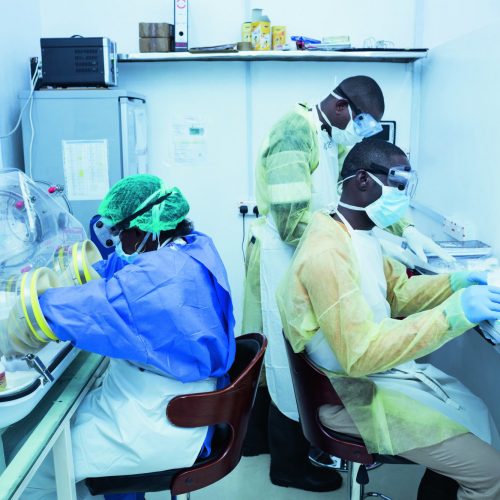
The clinical trial for Favipiravir, an antiviral medicine, which began under the direction of INSERM, is a multicenter non-randomized trial in which all the patients presented no contraindications when receiving Favipiravir with medical care.
126 patients infected with the Ebola virus voluntary participated in the study. “We quickly learned how to implement a clinical trial with patient participation, and this in the middle of an epidemic, in difficult circumstances”, continues Dr. Bing. The objective was to test the feasibility and acceptability of a trial during an Ebola outbreak, to gather data on the safety and efficacy of Favipiravir in reducing mortality and viral load in patients infected with the Ebola virus.
“The lessons learned during this study will surely help us to better elaborate therapeutic protocols based on the same type of molecules and to better care for patients” concludes Dr. Bing.
The Ebola Epidemic in West Africa, the most severe outbreak since the virus’ identification in Central Africa in 1976, began in 2013 in Guinea. According to the World Health Organization (WHO), it claimed the lives of more than 11,000 people, out of 28,000 identified cases.
In Guinea, ALIMA supported health authorities in the treatment of patients infected with the Ebola virus in N’Zérékoré.
The organization continues to provide medical care and psychosocial support to nearly 115 recovered patients.
On October 12, 2015, ALIMA received an awarded from the European Union for their actions in the fight against the Ebola virus.
Photo credit: Sylvain Cherkaoui / COSMOS / ALIMA